arts
What Colour does universal indicator turn in alkali?
Written by Emma Terry — 0 Views
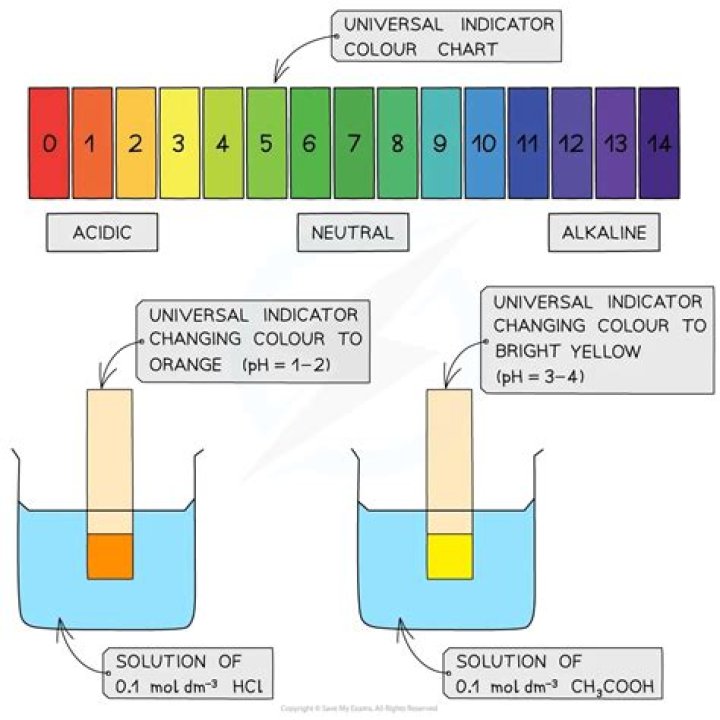
Universal indicator has many different colour changes, from red for strongly acidic solutions to dark purple for strongly alkaline solutions. In the middle, neutral pH 7 is indicated by green.
Similarly, it is asked, what Colour does universal indicator turn in a weak alkali?
Universal indicator
| pH range | Description | Colour |
|---|---|---|
| < 3 | Strong acid | Red |
| 3–6 | Weak acid | orange or yellow |
| 7 | Neutral | Green |
| 8–11 | Weak alkali | Blue |
Secondly, what Colour does universal indicator turn in sodium hydroxide? violet
Considering this, what Colour does universal indicator turn in base?
purple
What Colour does phenolphthalein turn in alkali?
Pink
Related Question Answers
Can you drink universal indicator?
Ingestion: May cause central nervous system depression, kidney damage, and liver damage. May cause gastrointestinal irritation with nausea, vomiting and diarrhea.Why is litmus purple in neutral solutions?
Hey! In general, solutions with a pH value of below 7 are acidic and those with more then pH 7 are basic/alkaline. According to the question,red litmus paper turns purple in the presence of a neutral solution. This means that the solution has pH value 7.Why is universal indicator not used in titrations?
A universal indicator will only give you relatively slow gradual colour changes, that are not precise enough to determine an end point accurately. Industry has long moved away from indicator end points in titration, where possible, and tends to favour potentiometric detection via automated instruments.Which one is correct for a universal indicator?
A universal indicator is a mixture of several indicators which are used to determine that the solution is acidic or basic. The strength of acid or base can be checked as the color change occurs in the indicator. Different color can be seen with the difference in the hydrogen ion concentration.What does blue mean on the pH scale?
The pH scale runs from 0 to 14, with each number assigned a different color. At the bottom of the scale sits red, which represents the most acidic, and a dark blue at its opposite end represents 14 and alkalinity. In the middle zone, the pH scale becomes neutral.What Colour does phenolphthalein turn in base?
pinkWhat Colour does lemon juice turn universal indicator?
In the lemon juice experiment, the pH paper turns from blue to vivid red, indicating the presence of H3O+ ions: lemon juice is acidic. Refer to the table of Universal Indicator Color change (figure 1 in the introduction) for clarification.What Colour does vinegar turn in universal indicator?
Answer: Universal indicator is a chemical that changes color in the presence of acids and bases from a pH of 2 to 10. Acids turn the indicator red, pink, orange, and yellow, while bases turn it green, blue, and purple. Vinegar is an acid, so when you poured the indicator solution into the second flask, it turned red.What is the advantage of universal indicator?
The advantage of the universal indicator is that it displays a variety of colors over the whole pH scale. This makes it possible to determine an approximate pH of a solution over a range from 1-14. whereas, A commonly used acid-base indicator is litmus.What are universal indicator why it is used?
The indicator is used to determine acidity or alkalinity of various substances such as soil, sewage, sludge, food etc. Solutions and liquids such as water, waste water, etc. Universal indicator is used more than other indicators because it has a wide range of results.What are the limits of the universal indicator?
Universal IndicatorIt covers a pH range from less than 3 (red) to greater than 11 (violet). Intermediate colors include orange/yellow (pH 3 to 6), green (pH 7 or neutral), and blue (pH 8 to 11).
Which substance will change the Colour of universal indicator from purple to red?
The Universal Indicator Color Guide shows that Universal Indicator turns red when it is added to a strong acid, it turns purple when it is added to a strong base, and it turns a yellowish-green when it is added to a neutral solution. All acids in the range of 1 to 4 turn the indicator red.What is a universal indicator Class 10?
To measure the strength of an acid or a base solution we use universal indicators. Universal indicator is a mixture of many indicators which gives diferent colours at different pH values of entire scale. It shows different colours at different concentration of ions in the solution. pH. colour.Is pH paper a universal indicator?
The major difference between the pH paper and universal indicator is the pH paper gives the exact pH of the substance, whereas the universal indicator used to find out the major difference between the pH paper and universal indicator is the pH paper gives the exact pH of the substance, whereas the universal indicatorWhat does purple mean on the pH scale?
First, high levels of chlorine (greater than 15 ppm) or bromine (greater than 20 ppm) can cause the pH indicator to turn purple, which does not match the pH color scale. This is similar to the high pH colors, 8.0 to 8.4 ppm, so be careful not to mistake it for a high reading and then add acid when it's not necessary.What is the Colour of magnesium sulphate on universal indicator?
Answer: Magnesium Sulphate being an acid,shows the color between red and Yellow on testing with the universal indicator or the pH paper.What happens when sodium reacts with water and universal indicator?
When the sodium metal is reacting with the water, the sodium metal goes to the ion state and becomes a stable element. The universal indicator reacts with the sodium hydroxide, which is a base that makes the indicator turn from green to purple.What gas makes universal indicator turn red?
AmmoniaWhat Colour is universal indicator in ammonia?
Task 1: Using universal indicator| Chemical | Colour | pH |
|---|---|---|
| Potassium hydroxide | Answer navy - very dark blue | Answer ~ 14 |
| Sodium hydroxide | Answer navy - very dark blue | Answer ~ 14 |
| Ammonia solution | Answer dark blue | Answer ~ 11.5 |
| Water | Answer green | Answer ~ 7 |
Why does the pink color disappear in a titration?
Phenolphthalein is an acid/base indicator. It is colorless when it is an acid and pink when it is a base. When enough carbonic acid is formed it neutralizes the lime water, which is a base and makes the solution an acid. Therefore with the phenolphthalein, the color disappears in the now acidic solution.Why phenolphthalein is pink in basic medium?
Ionization and the Phenolphthalein IndicatorPhenolphthalein is a weak acid and is colorless in solution although its ion is pink. Adding hydroxide ions (OH-, as found in bases) will change the phenolphthalein into its ion and turn the solution pink.