What is gold heap leaching?
Accordingly, what is heap leaching process?
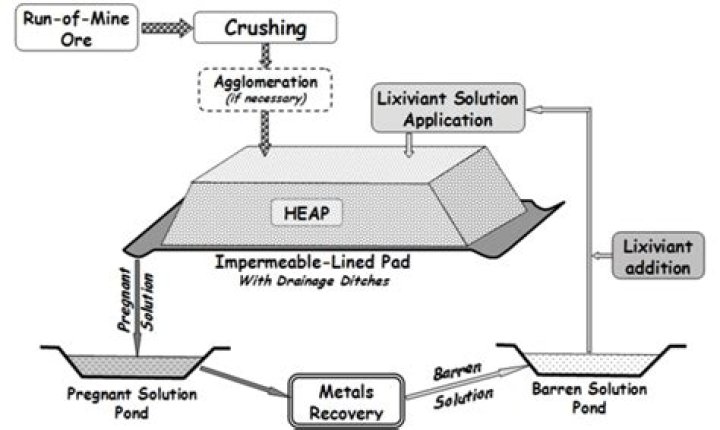
Heap leaching is an industrial mining process used to extract precious metals, copper, uranium, and other compounds from ore using a series of chemical reactions that absorb specific minerals and re-separate them after their division from other earth materials.
Likewise, what does leaching mean in mining? Leaching is a process widely used in extractive metallurgy where ore is treated with chemicals to convert the valuable metals within into soluble salts while impurity remain insoluble. These can then be washed out and processed to give the pure metal; the material left over is commonly referred to as tailings.
Just so, why is cyanide heap leaching extracted gold?
Cyanide can be used to extract gold, either in a controlled mill environment, or more crudely on rock piles in the open. Cyanide "vat leaching" mixes finely crushed ore with a cyanide salt in water. The cyanide binds to the gold ions, and makes them soluble in water, thereby allowing separation from the rock.
What is agitation leaching?
Agitation leaching generally involves dispersing the finely ground ore in water to obtain a pulp containing 40–70% by weight of the solids and subsequently adding the acid or base solution.
Related Question Answers
What are leaching?
In agriculture, leaching is the loss of water-soluble plant nutrients from the soil, due to rain and irrigation. Leaching may also refer to the practice of applying a small amount of excess irrigation where the water has a high salt content to avoid salts from building up in the soil (salinity control).What is ore deposit?
An ore deposit is a natural concentration of one or more minerals within the host rock. It has a definite shape on economic criteria with finite quantity (tons) and average quality (grade). The shape varies according to the complex nature of the deposit such as layered, disseminated, veins, folded, and deformed.What does Lixiviant mean?
A lixiviant is a liquid medium used in hydrometallurgy to selectively extract the desired metal from the ore or mineral. It assists in rapid and complete leaching, for example during in situ leaching. The metal can be recovered from it in a concentrated form after leaching.What is meant by metallurgy?
noun. the technique or science of working or heating metals so as to give them certain desired shapes or properties. the technique or science of making and compounding alloys. the technique or science of separating metals from their ores.What is bioleaching chemistry?
Bioleaching is the extraction of metals from their ores through the use of living organisms. Bioleaching is one of several applications within biohydrometallurgy and several methods are used to recover copper, zinc, lead, arsenic, antimony, nickel, molybdenum, gold, silver, and cobalt.How gold is extracted by cyanide process?
cyanide process or cyanidation, method for extracting gold from its ore. The ore is first finely ground and may be concentrated by flotation; if it contains certain impurities, it may be roasted. It is then mixed with a dilute solution of sodium cyanide (or potassium or calcium cyanide) while air is bubbled through it.Is cyanide used in gold making?
A sodium cyanide solution is commonly used to leach gold from ore. The cyanide dissolves the gold from the ore into the solution as it trickles through the heap. The pad collects the now metal-impregnated solution which is stripped of gold and resprayed on the heap until the ore is depleted.How does gold leaching work?
Leaching dissolves the gold out of the ore using a chemical solvent. As the cyanide and oxygen react chemically, gold in the pulp dissolves. When workers introduce small carbon grains to the tank, the gold adheres to the carbon. Filtering the pulp through screens separates the gold-bearing carbon.How do you recover gold from gold potassium cyanide?
A process is described involving gold in which gold cyanide ion (e.g., potassium gold cyanide) is removed from an aqueous solution (typically a water rinse solution) by absorption on an anion-exchange resin and the gold cyanide ion is then removed from the resin using an eluent solution comprising organic solvent,Where is cyanide found?
Where cyanide is found and how it is used. Cyanide is released from natural substances in some foods and in certain plants such as cassava, lima beans and almonds. Pits and seeds of common fruits, such as apricots, apples, and peaches, may have substantial amounts of chemicals which are metabolized to cyanide.Why do Jewellers use cyanide?
Cyanide is used in the extraction and polishing of gold, and for gold-plating. The ornament industry uses the chemical to give gold its reddish yellow colour, believed to be the “original” colour of the metal, and for ridding it of impurities.Is cyanide illegal?
Possessing sodium cyanide is not illegal because it is used in mining to extract gold and for other industrial purposes.What is ore of gold?
Gold - The primary mineral of gold is the native metal and electrum (a gold-silver alloy). Some tellurides are also important ore minerals such as calaverite, sylvanite, and petzite. Hafnium - Primary ore mineral is zircon. Indium - Primarily is a byproduct of zinc processing.Can cyanide dissolve gold?
Gold and silver are not soluble in water. A complexant, such as cyanide, which stabilizes the gold and/or silver species in solution, and an oxidant such as oxygen are required to dissolve these metals. The amount of cyanide in solution required for dissolution may be as low as 350 mg/l or 0.035% (as 100% NaCN).How gold is mined step by step?
The Mining Process- Mining – open pit and underground. To define the ore from the waste rock, samples are taken and assayed.
- Crushing.
- Transport.
- Grinding and sizing.
- Leaching and adsorption.
- Elution and electrowinning.
- Bullion production.
- Water treatment.
How does leach mining work?
Heap leaching is an industrial mining process used to extract precious metals, copper, uranium and other compounds from ore using a series of chemical reactions that absorb specific minerals and re-separate them after their division from other earth materials.Where is situ leaching used?
It has been used in the US state of Colorado to extract nahcolite (sodium bicarbonate). In-situ leaching is often used for deposits that are too deep, or beds that are too thin, for conventional underground mining.How does solution mining work?
Solution mining refers to the production of salt (or potash, or other soluble products) by pumping water into subterranean salt deposits, found in many parts of the world, dissolving the salts and pumping the brine to the surface for drying and further use. In the cavern, the hydrocarbons lie on top of the brine.What is the process of underground mining?
Underground miningUnderground mining is used to extract ore from below the surface of the earth safely, economically and with as little waste as possible. The entry from the surface to an underground mine may be through a horizontal or vertical tunnel, known as an adit, shaft or decline.