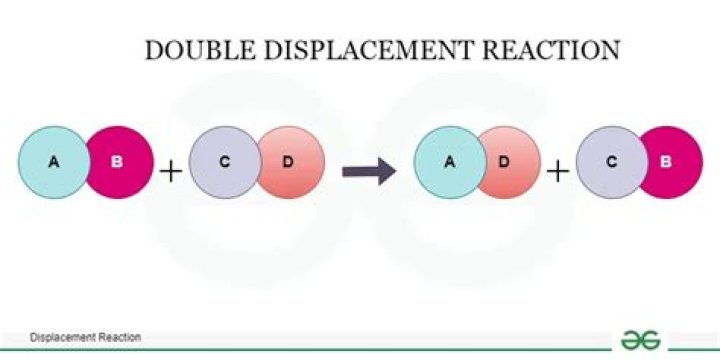
The double-displacement reaction generally takes the form of AB + CD → AD + CB where A and C are positively-charged cations, while B and D are negatively-charged anions. In a double-displacement reaction, one of the products is typically a precipitate, a gas, or a molecular compound. Consequently, what is the equation of displacement reaction?
A single-displacement reaction, is a chemical reaction in which one element is replaced by another in a compound. It can be represented generically as: A + B-C → A-C + B.
Also, what is a double displacement reaction example? Double displacement reactions generally take place in aqueous solutions in which the ions precipitate and there is an exchange of ions. For example, on mixing a solution of barium chloride with sodium sulphate, a white precipitate of barium sulphate is immediately formed. These reactions are ionic in nature.
One may also ask, what is the equation of displacement and double displacement reaction?
When solutions of sodium carbonate and hydrochloric acid are mixed, the equation for the hypothetical double displacement reaction is: Na2CO3 + 2 HCl → 2 NaCl + H2CO3 Bubbles of a colorless gas are evolved when these solutions are mixed.
What is a double displacement reaction Class 10?
Answer: Those reactions in which two compounds react by an exchange of ions to form two new compounds are called double displacement reactions.
Related Question Answers
What is double displacement reaction Class 10 Brainly?
Textbook solutionThis is the type of chemical change in which two compounds in a solution react to form two new compounds by mutual exchange of radicals is known as double displacement reactions. This reaction has two types i.e. precipitation reaction and neutralization reaction.
What is double replacement in chemistry?
Double replacement reactions—also called double displacement, exchange, or metathesis reactions—occur when parts of two ionic compounds are exchanged, making two new compounds. What is double decomposition reaction in chemistry?
Definitions of double decomposition reaction. a chemical reaction between two compounds in which parts of each are interchanged to form two new compounds (AB+CD=AD+CB) What is double displacement reaction Class 9?
Double displacement reactions may be defined as the chemical reactions in which one component each of both the reacting molecules is exchanged to form the products. During this reaction, the cations and anions of two different compounds switch places, forming two entirely different compounds. Is double displacement a displacement reaction?
A double-replacement (or double-displacement) reaction is a reaction in which the positive and negative ions of two ionic compounds exchange places to form two new compounds. Double-replacement reactions can form precipitates, gases, or molecular compounds. What is displacement and double displacement reaction Class 10th?
When a reactive element displaces a less reactive one from a molecule and takes its place, it is called displacement reaction. When an interchange of two atoms from two different compounds takes place and the product contains totally different compounds, it is a double displacement reaction. What is displacement reaction in Ncert?
In displacement reaction a more reactive metal displaces a less reactive metal but a less rwactive metal cannot displace a more reactive metal. In beaker A Zn being more reactive than copper replaces copper from the solution of copper sulphate and forms zinc sulphate. Which option denotes a double displacement reaction?
A double replacement reaction is represented by the general equation. In a double displacement reaction, atoms from two different compounds switch places. The reactants are two compounds and the products are two different compounds. Double displacement reactions can also be called double replacement reactions. What is a displacement reaction Class 8?
A more reactive metal displaces a less reactive metal from its salt solution. When a more reactive metal is placed in the salt solution of a less reactive metal,then the more reactive metal displaces the less reactive metal from its salt solution. What is a precipitate Class 10?
Hint:Precipitate is the insoluble solid which settles down after the completion of the chemical reaction. The formation of the precipitate can help in determining the presence of various types of ions or atoms. What is double displacement reaction write 5 examples?
AgNO 3 + NaCl → AgCl + NaNO. 2NaCl + CaSO4 → Na2SO4+ CaCl. H2SO4 + 2LiOH ⇌ Li2SO4 + 2H2O. AgNO3 + HCl ⇌ AgCl + HNO. What is the decomposition equation?
A decomposition reaction occurs when one reactant breaks down into two or more products. It can be represented by the general equation: AB → A + B. In this equation, AB represents the reactant that begins the reaction, and A and B represent the products of the reaction. Which of the following is a balance chemical equation?
A balanced chemical equation occurs when the number of the atoms involved in the reactants side is equal to the number of atoms in the products side. In this chemical reaction, nitrogen (N2) reacts with hydrogen (H) to produce ammonia (NH3). What is the general formula for a decomposition reaction?
A decomposition reaction occurs when one reactant breaks down into two or more products. This can be represented by the general equation: AB → A + B. Examples of decomposition reactions include the breakdown of hydrogen peroxide to water and oxygen, and the breakdown of water to hydrogen and oxygen. What is double replacement example?
An example of a double replacement reaction is the reaction between silver nitrate and sodium chloride in water. The silver ion picks up the sodium's chloride ion to form silver chloride, while the sodium ion picks up the nitrate anion to form sodium nitrate. Like the reactants, both products are ionic compounds.