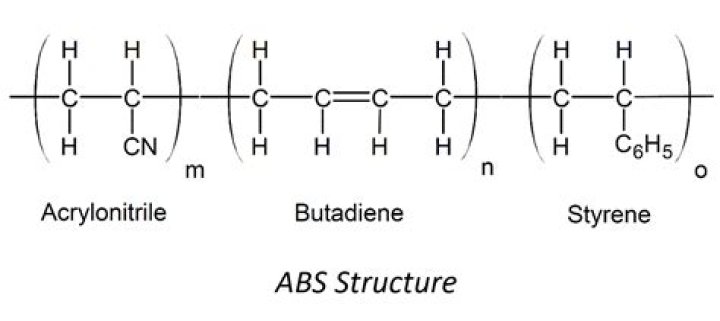
What is the structure of acrylonitrile?
Furthermore, what is the Iupac name of acrylonitrile?
| IUPAC Name | prop-2-enenitrile |
|---|---|
| Alternative Names | ACRYLONITRILE 2-Propenenitrile Vinyl cyanide |
| Molecular Formula | C3H3N |
| Molar Mass | 53.064 g/mol |
| InChI | InChI=1S/C3H3N/c1-2-3-4/h2H,1H2 |
Likewise, is Acrylonitrile an alkene? Acrylonitrile actually has two important functional groups, one being an alkene and the other a nitrile. An alkene is an organic functional group that contains two carbon atoms joined together by a carbon-carbon double bond.
Also asked, is acrylonitrile a monomer?
Acrylonitrile is used principally as a monomer to prepare polyacrylonitrile, a homopolymer, or several important copolymers, such as styrene-acrylonitrile (SAN), acrylonitrile butadiene styrene (ABS), acrylonitrile styrene acrylate (ASA), and other synthetic rubbers such as acrylonitrile butadiene (NBR).
Is made from a chemical called acrylonitrile?
Acrylonitrile is used to make other chemicals such as plastics, synthetic rubber, and acrylic fibers.
Related Question Answers
Is acrylonitrile and acrylic same?
is that acrylonitrile is (organic compound) a toxic colourless liquid organic compound, ch2=chcn, synthesized from propylene and ammonia; used as a monomer in the production of acrylic resins and synthetic rubber while acrylic is (organic chemistry) an acrylic resin.How can acrylonitrile be prevented?
Mixtures of Acrylonitrile with air are flammable. In its liquid state, Acrylonitrile has a tendency to polymerise, which can however be prevented by the addition of phenolic (e.g. methyl ether of hydroquinone or amine-based stabilizers and small quantities of water.What is the hybridization of acrylonitrile?
It is useful to remember (In future chemistry classes as well) that carbons with one double bond to it is sp2 hybridized, and is sp hybridized if attached to a triple bond. Thus, H2C=CHC N, first carbon starting from the left is sp2, second carbon is sp2, third carbon is sp, and the nitrogen is sp.What is acrylonitrile used in?
Acrylonitrile is used in many industries. It is used to make certain plastics, rubbers, and chemicals, and in the past, as a pesticide. Some examples of workers at risk of being exposed to acrylonitrile include the following: Workers involved in the manufacturing of acrylic fibers and plastics.Is polyacrylonitrile toxic?
Ingestion: May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated. Metabolism may release cyanide, which may result in headache, dizziness, weakness, collapse, unconsciousness and possible death.What elements make up acrylonitrile?
Acrylonitrile is a synthetic monomer produced from propylene and ammonia; butadiene is a petroleum hydrocarbon obtained from the C4 fraction of steam cracking; styrene monomer is made by dehydrogenation of ethyl benzene — a hydrocarbon obtained in the reaction of ethylene and benzene.Is a polymerised acrylonitrile?
Acrylonitrile polymers (also called nitriles) are copolymers of acrylonitrile and various other monomers. The three most important comonomers are styrene, butadiene, and methyl acrylate. About 30 to 85 percent by weight of the copolymer is acrylonitrile and about 15 percent or more is butadiene and/or methyl acrylate.What is the monomer unit of natural rubber?
isopreneHow do you make acrylonitrile?
Acrylonitrile is manufactured by combining propylene, ammonia, and air in a process called ammoxidation. During ammoxidation, propylene, ammonia and air are fed through a catalyst at a high temperature.Is Buna S synthetic rubber?
Note: BUNA-S is going to form from its monomeric units (Styrene and butadiene) through addition polymerization reaction in the presence of Sodium. It is not a natural rubber, it is a synthetic rubber.Which polymer is formed by Chloroethene?
vinyl chlorideWhat is the monomer of nylon 6?
caprolactamWhat does acrylonitrile smell like?
Acrylonitrile is a clear, colorless or slightly yellow liquid that is highly volatile and toxic. Acrylonitrile vapor is heavier than air. It has a pungent odor of onion or garlic that does not provide adequate warning of hazardous levels. Acrylonitrile is poisonous by inhalation, ingestion or skin contact.Which is a polymer of acrylonitrile?
polyacrylonitrile (PAN), a synthetic resin prepared by the polymerization of acrylonitrile. A member of the important family of acrylic resins, it is a hard, rigid thermoplastic material that is resistant to most solvents and chemicals, slow to burn, and of low permeability to gases.What is Chloroethene?
Chloroethene is a man-made substance with a mild, sweet odor. Chloroethene exists in a liquid form if kept under high pressure or at low temperatures. Choroethene is a colorless gas at room temperature. It burns easily.Is propionitrile an alkyne?
Propionitrile, also known as ethyl cyanide and propanenitrile, is an organic compound with the formula CH3CH2CN. It is a simple aliphatic nitrile. It is used as a solvent and a precursor to other organic compounds.What acrylonitrile does to the body?
* Exposure to Acrylonitrile can cause weakness, headache, dizziness, confusion, nausea, vomiting, and can lead to death. * Repeated exposure can irritate the nose causing discharge, nosebleeds, and sores inside the nose. * Acrylonitrile may affect the liver.What is the formula of styrene?
C8H8Is hydrogen a cyanide?
Hydrogen cyanide, sometimes called prussic acid, is a chemical compound with the chemical formula HCN. It is a colorless, extremely poisonous, and flammable liquid that boils slightly above room temperature, at 25.6 °C (78.1 °F).Hydrogen cyanide.
| Names | |
|---|---|
| ChemSpider | 748 |
| ECHA InfoCard | 100.000.747 |
| EC Number | 200-821-6 |
| KEGG | C01326 |
Does acrylonitrile react with water?
Environmental Fate and Behavior Acrylonitrile is both readily volatile in air and highly soluble in water. It is oxidized by strong oxidants such as chlorine used to disinfect water. Acrylonitrile is readily degraded by aerobic microorganisms in water.What is the mass in grams of acrylonitrile?
mass of Acrylonitrile (C3H3N) = 53.06 g/mol * 0.209 mol = 11.072 g.What is the formula of acetonitrile?
C2H3NIs Orlon a polymer?
Orlon is the name that is used in trade purposes for Polyacrylonitrile (PAN). It is a polymer of acrylonitrile which is commonly known as vinyl cyanide (VCN). Therefore, option D is correct. The chemical formula of vinyl cyanide is CH2CHCN.What is the percent composition of acrylonitrile?
The percent composition of carbon in acrylonitrile is a 5.690%What is polyacrylonitrile made of?
polyacrylonitrile (PAN), a synthetic resin prepared by the polymerization of acrylonitrile. A member of the important family of acrylic resins, it is a hard, rigid thermoplastic material that is resistant to most solvents and chemicals, slow to burn, and of low permeability to gases.Is acetonitrile acidic or basic?
Acetonitrile| Names | |
|---|---|
| Acidity (pKa) | 25 |
| UV-vis (λmax) | 195 nm |
| Absorbance | ≤0.10 |
| Magnetic susceptibility (χ) | −28.0×10−6 cm3/mol |