Two factors determine whether a substance is a solid, a liquid, or a gas: The kinetic energies of the particles (atoms, molecules, or ions) that make up a substance. Kinetic energy tends to keep the particles moving apart. The attractive intermolecular forces between particles that tend to draw the particles together. Keeping this in view, what does a state of matter depend on?
These are the three common states of matter. Whether a substance is a solid, a liquid, or a gas depends on its temperature and the pressure placed on it. At room temperature (about 22°C) and at the normal pressure exerted by the atmosphere, water exists as a liquid, which can flow from one container to another.
Also Know, what determines a substance's state of matter at a given temperature? Explain what determines a substance's state at a given temperature. The type of force and it's strength determines a substance's state of a given temperature. Intermolecular forces are forces between molecules well intra-molecular forces are forces within molecules. They both determine a molecule shape.
Also asked, which is the most important factor in determining the state of a substance quizlet?
Discuss why temperature is the most important factor in determining the state of a substance. Compare and contrast gases and plasmas. Both gases and plasmas consist of energetic particles that are so far apart that attractive forces between particles are inconsequential.
How is the plasma state of a substance different from the gas of the same substance?
It takes a very special environment to keep plasmas going. They are different and unique from the other states of matter. Plasma is different from a gas, because it is made up of groups of positively and negatively charged particles. In neon gas, the electrons are all bound to the nucleus.
Related Question Answers
What are the 26 states of matter?
The classical states of matter are usually summarized as: solid, liquid, gas, and
plasma.
Low-energy states
- Solid: A solid holds a definite shape and volume without a container.
- Liquid: A mostly non-compressible fluid.
- Gas: A compressible fluid.
What are the 22 states of matter?
Here is the list I will provide: - Solid.
- Liquid.
- Gas.
- Plasma.
- Bose-Einstein Condensate.
- Excitonium.
- Degenerate Matter.
- Photonic Matter.
What is the fifth state of matter?
The fifth state of matter is the Bose-Einstein condensate. In 1920, Indian physicist Satyendra Nath Bose had done some calculations for the fifth state of matter. Albert Einstein predicted a new state of matter — the Bose-Einstein condensate (BEC). What is it called when matter changes state?
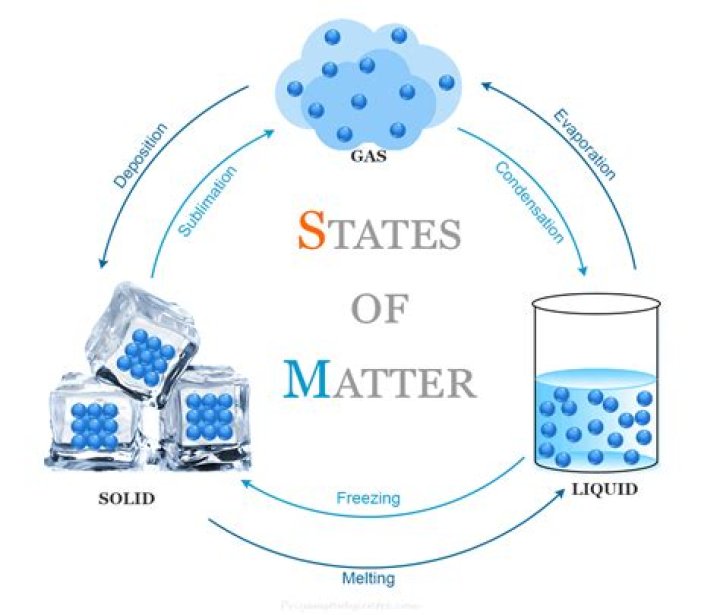
Lesson Summary. Changes of state are physical changes. They occur when matter absorbs or loses energy. Processes in which matter changes between liquid and solid states are freezing and melting. Processes in which matter changes between liquid and gaseous states are vaporization, evaporation, and condensation. What are the 18 states of matter?
The classical states of matter are usually summarized as: solid,
liquid, gas, and
plasma.
Low-energy states
- Solid: A solid holds a definite shape and volume without a container.
- Liquid: A mostly non-compressible fluid.
- Gas: A compressible fluid.
How do we classify matter?
Matter can be classified according to physical and chemical properties. Matter is anything that occupies space and has mass. The three states of matter are solid, liquid, and gas. A physical change involves the conversion of a substance from one state of matter to another, without changing its chemical composition. Is fire a plasma?
Fire (flame) Fire (flames) may contain plasma, albeit one that is a partially ionized plasma, and dominated by collisions: Any ionized gas cannot be called a plasma, of course; there is always a small degree of ionization in any gas. What is the most important factor in determining the state of a substance?
Discuss why temperature is the most important factor in determining the state of a substance. Which type of reaction is always spontaneous quizlet?
True or False: Exergonic reactions are always spontaneous and endergonic reactions are always non-spontaneous. For the process H2O (g) → H2O (l), ΔH is negative and ΔS is negative. What two factors determine the physical state of a substance?
Two factors determine whether a substance is a solid, a liquid, or a gas: The kinetic energies of the particles (atoms, molecules, or ions) that make up a substance. Kinetic energy tends to keep the particles moving apart. The attractive intermolecular forces between particles that tend to draw the particles together. What are the two main classes of matter and what characteristics differentiate them?
Matter can be broken down into two categories: pure substances and mixtures. Pure substances are further broken down into elements and compounds. Mixtures are physically combined structures that can be separated into their original components. A chemical substance is composed of one type of atom or molecule. What are the two classification of matter?
Two principal ways of classifying matter are according to its physical state (as a gas, liquid, or solid) and according to its composition (as an element, compound, or mixture). States of Matter. A sample of matter can be a gas, a liquid, or a solid. These three forms of matter are called the states of matter. What main factor determines a materials State of Matter How does it affect the particles of a substance?
Two factors determine whether a substance is a solid, a liquid, or a gas: The kinetic energies of the particles (atoms, molecules, or ions) that make up a substance. Kinetic energy tends to keep the particles moving apart. The attractive intermolecular forces between particles that tend to draw the particles together. How does temperature affect the movement of particles?
When heat is added to a substance, the molecules and atoms vibrate faster. As atoms vibrate faster, the space between atoms increases. The motion and spacing of the particles determines the state of matter of the substance. The end result of increased molecular motion is that the object expands and takes up more space. Which properties can you use to distinguish between matter and Nonmatter?
Matter has mass, occupies a measurable volume, and can be sensed and measured. Nonmatter has no mass and does not occupy a measurable volume (although it can occupy space); nonmatter can be sensed (often with instruments) and measured. Light energy is a form of nonmatter studied by scientists. What is all matter made up of?
Idea a: All matter is made up of particles called atoms and molecules (as opposed to being continuous or just including particles). On the following page, the idea is stated as one of four concepts in Dalton's theory: “All matter is composed of tiny, indivisible particles called atoms” (p. 158s). Is h2 a hydrogen bond?
Answer and Explanation: H2 is not a hydrogen bond but is a molecule in which hydrogen is bonded to itself. H2 forms when two hydrogen atoms, H, are bonded together by a What is the chief factor that determines the physical state of matter?
The chief factor responsible for the physical state of the sample of matter is to be explained. Concept Introduction: Matter- Anything that has mass and that can occupy some space is termed as the matter. What is the most common state of matter in the universe?
The funny thing about that is, that as far as we know, plasmas are the most common state of matter in the universe. They are even common here on earth. A plasma is a gas that has been energized to the point that some of the electrons break free from, but travel with, their nucleus. What is the difference between intramolecular and intermolecular?
Intramolecular forces are the forces that hold atoms together within a molecule. Intermolecular forces are forces that exist between molecules. What gives each state of matter its unique properties?
Solid is the state in which matter maintains a fixed volume and shape; liquid is the state in which matter adapts to the shape of its container but varies only slightly in volume; and gas is the state in which matter expands to occupy the volume and shape of its container. How does surface tension depend on intermolecular forces?
Surface tension is caused by the effects of intermolecular forces at the interface. Surface tension depends on the nature of the liquid, the surrounding environment and temperature. Liquids were molecules have large attractive intermolecular forces will have a large surface tension. Can solids and liquids be compressed?
In most liquids, the particles are less densely packed, giving them the ability to move around and slide past each other. While a liquid is easier to compress than a solid, it is still quite difficult – imagine trying to compress water in a confined container! What causes surface tension?
Surface Tension: "The property of the surface of a liquid that allows it to resist an external force, due to the cohesive nature of its molecules." The cohesive forces between liquid molecules are responsible for the phenomenon known as surface tension. What is the surface tension and what conditions must exist for it to occur?
What conditions must exist for surface tension to occur? 1. the stronger the attraction between particles the stronger the attraction. Explain why the surface of water in a graduated cylinder is curved. What is the purest form of matter?
An element is the simplest form of matter which cannot be split into two or more simpler substances by ordinary chemical methods because an element is formed of only one type of atom. What is common in the three states of matter?
The common thing among the three states of matter is - They are made up of small tiny particles. They have a particular mass and can occupy space . This three states have volume in it . The atoms of this three states have force of attractions between them . What are the 15 states of matter?
Currently voted the best answer. Less familiar phases include: quark-gluon plasma; Bose-Einstein condensates and fermionic condensates; quantum spin Hall state; degenerate matter; strange matter; superfluids and supersolids; and possibly string-net liquids. Why is blood plasma called plasma?
Plasma, also called blood plasma, the liquid portion of blood. Plasma serves as a transport medium for delivering nutrients to the cells of the various organs of the body and for transporting waste products derived from cellular metabolism to the kidneys, liver, and lungs for excretion. How many types of matter are there?
There are four natural states of matter: Solids, liquids, gases and plasma. The fifth state is the man-made Bose-Einstein condensates. In a solid, particles are packed tightly together so they don't move much. Can plasma be compressed?
Gaseous substances will then easily compress, resulting in lots of heating as they do, until they heat up enough that the electrons freely float among the nuclei, and you have just made a Plasma. More compression, resulting in more heat. What are the two types of pure substances?
There are two types of pure substances that are Elements and Compounds. Examples of elements are: Iron, Silver, Gold, Mercury etc. Examples of compounds are: Water, Carbon dioxide, methane, vinegar etc. Is the sun made of plasma?
So, although it is common to see the Sun referred to as a gaseous body, a more specific description is that it is made of plasma. In this case, the plasma consists of hydrogen and helium ions, together with the electrons that were liberated when those ions were produced.” What is plasma state of matter in Hindi?
?????? ?? ????? ??????? ???, ???????? ????? ??? ?? ??????? ?? ??? ??, ?????? ???????????? ?? ?? ??????? ?????? ???? ?????? ?? ??? ?? ??? ???? ???? ?? ???? ???????? ???? ??? ???????? ?? ??? ???, ???? ?? ??? ?? ????? ?? ???? ?????? ??? ?? ????? ??? ?????? ?? ?? ????? ?????? ???? ???? ???