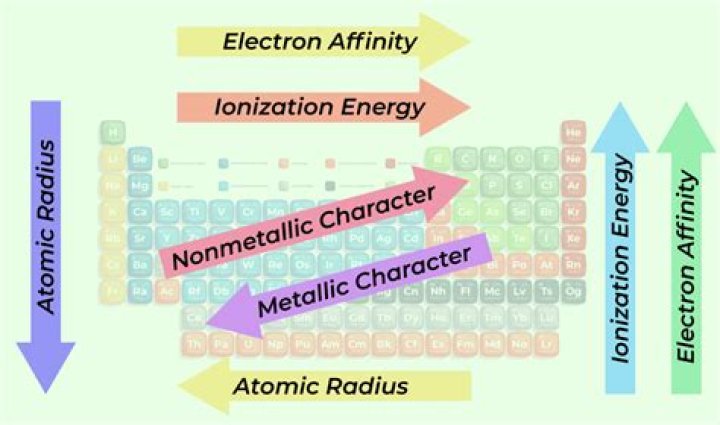
Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. This makes the fluoride anion so formed unstable (highly reactive) due to a very high charge/mass ratio. As a result, fluorine has an electron affinity less than that of chlorine. Also asked, does F or Cl have higher electron affinity?
Electron affinity of fluorine is less than that of chlorine. This is due to the reason explained below: Fluorine has seven electrons in 2p-subshell whereas chlorine has seven electrons in its 3p-subshell. Therefore, repulsion among the electrons will be more in the 2p-shell of fluorine than 3p-subshell in chlorine.
Additionally, which F Cl has the highest electron affinity and why? Now coming to the question, as we have already seen; Br has the lowest electron affinity because of high atomic size and therefore has less tendency to accept electrons to form anion whereas Cl has highest electron affinity because of less atomic size and has high tendency to accept electrons.
In this regard, which is more electronegative F or Cl?
At the bottom left corner of the periodic table lies Cesium, which lacks a catchy nickname [but has attracted a strangely devoted following] with an electronegativity of 0.79.
Carbon is More Electronegative Than You Think.
| Element | Electronegativity (Pauling) |
| F | 4.0 [3.98] |
| O | 3.4 [3.44] |
| Cl | 3.2 [3.16] |
| N | 3.0 [3.04] |
Does CL have electron affinity?
The first electron affinity is the energy released when 1 mole of gaseous atoms each acquire an electron to form 1 mole of gaseous -1 ions. First electron affinities have negative values. For example, the first electron affinity of chlorine is -349 kJ mol-1. By convention, the negative sign shows a release of energy.
Related Question Answers
What element has highest affinity?
Therefore, chlorine has a higher electron affinity than fluorine, and this orbital structure causes it to have the highest electron affinity of all of the elements. Which element has the highest most positive electron affinity?
Chlorine
What are the exceptions to electron affinity?
In general, exceptions arise when new subshells are being filled/half-filled, or in cases where the atom is too small. In the first case, Be and Mg are interesting examples: they have a positive electron affinity (just like N, in fact) because of the energy difference between the s and p subshells. Why is Cl more EA than F?
the electron affinity of the fluorine is less than chlorine because the size of fluorine is too small as size decreases from left to right inside period, whereas chlorine has a larger size to accommodate electrons hence electron affinity of chlorine is more than fluorine. What is a positive electron affinity?
The electron affinity is defined as the energy change that occurs when an atom gains an electron, releasing energy in the process. If a reaction is exothermic, the change in energy is negative. This means that the electron affinity is positive. Which has the most negative electron affinity?
Chlorine has the most negative electron affinity. According to Wikipedia [Electron affinity] , In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as the amount of energy released when an electron is added to a neutral atom or molecule in the gaseous state to form a negative ion. Is Cl more electronegative than Br?
Atomic chlorine is much more electronegative than atomic bromine, which is more electronegative than atomic iodine. Which has the greatest electron affinity SE Cl or Br?
Chlorine does have a higher electron affinity than bromine. A significant amount of energy is released when an electron is added to these elements because this incoming electron allows them to complete their octet. Which of the following will have the least electron affinity 1 P 2 S 3 Cl 4 F?
The electron affinity for oxygen is lowest. Which halogen has the smallest electron affinity F Cl Br I?
An electron will not be as attracted to the nucleus, resulting in a low electron affinity. However, fluorine has a lower electron affinity than chlorine. This can be explained by the small size of fluorine, compared to chlorine. Which element has lowest electron affinity in oxygen family?
Thus, in case of oxygen and sulphur which are present in the same group, sulphur has greater electron affinity than oxygen. So, out of the given elements to us, nitrogen has the lowest electron affinity. Hence, the correct option is (A) Nitrogen. What is the correct order of electron affinity of oxygen family?
S > Se > O. Which one of the four ions has largest electron affinity?
F− ions
Which element has the highest electron affinity in oxygen family?
Fluorine. D. Oxygen. Hint: We should remember that the element which will release the most amount of energy on adding an electron in its isolated gaseous atom will posse's highest electron affinity in the periodic table. Why is electron affinity so positive for noble gas elements?
The noble gases present in the group 18 of periodic table, have electronic configuration ns2np6. Thus, in case of noble gases, where it resists the addition of electrons, extra energy is required in order to force the electron to bind to the stable atom. Therefore, making the electron gain enthalpies positive. Why do halogens have high electron affinity?
The high electron affinities of the halogens are a result of their having an almost complete outer shell of electrons. Because of this overall attraction, energy will be released when the electron is captured by the fluorine atom. Similar reasoning also explains why oxygen also has a high electron affinity. Which element of Group 17 has the highest electron affinity?
chlorine
What is the difference between electronegativity and electron affinity?
Electronegativity refers to the ability of the atoms to attract the electrons from the other elements. Electron affinity refers to the amount of energy that is liberated whenever a molecule or a neutral atom tends to acquire an electron from the other elements.