Which is adsorbed more on activated charcoal?

Also know, which is adsorbed to maximum amount by activated charcoal?
As CO2 is an easily liquefiable gas, hence it is adsorbed more by charcoal.
One may also ask, does activated charcoal absorb or adsorb? Activated charcoal (also called active carbon) is so effective at filtering because it works through the process of adsorption. In other words, a wide range of impurities can chemically adhere to its surface.
Keeping this in view, which will be adsorbed more readily on the surface of charcoal and why?
Solution : NH3 has higher critical temperature than CO2 , i.e., NH3 is more liquefiable than CO2 , Hence, NH3 has greater intermolecular forces of attraction and hence will be adsorbed more readily.
Which is maximum absorbed by a charcoal?
N2 will be strongly absorbed by charcoal because of more surface area and can be easily liquified.
Related Question Answers
Why is activated charcoal activated?
When you take activated charcoal, drugs and toxins can bind to it. This helps rid the body of unwanted substances. Charcoal is made from coal, wood, or other substances. It becomes "activated charcoal" when high temperatures combine with a gas or activating agent to expand its surface area.What is activated charcoal made of?
Activated charcoal is made by burning natural, carbon-rich materials (bamboo, wood, coconut shells, olive pits or coal) in low-oxygen concentrations. This process extracts the hydrogen, methane and tar from the material, reducing its weight and creating a black, mostly carbon substance.Which gas can be adsorbed more?
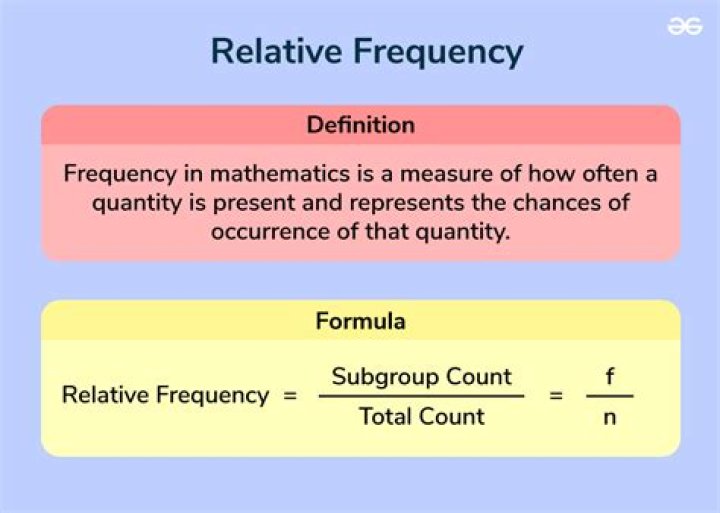
The phenomenon of adsorption of gases by solids depends upon: -Nature of adsorbent: Easily liquefiable gases such as NH3 , CH4 , HCI, CI2 , SO2 , CO etc. are adsorbed more than permanent gases like H2 , O2 , N2 etc.Which of the following gases is absorbed easily and more on activated charcoal?
Gases having higher critical temperature are easily liquefied. Hence in the liquid state they have greater force of attraction with the charcoal and therefore they are more easily adsorbed on the surface of the charcoal. Therefore, the gas which has the highest critical temperature is Methane or CH4.How many layers are adsorbed in chemical adsorption?
one layerWhat does desorption mean?
Desorption is a phenomenon whereby a substance is released from or through a surface. The process is the opposite of sorption (that is, either adsorption or absorption). In chemistry, especially chromatography, desorption is the ability for a chemical to move with the mobile phase.Why is adsorption is exothermic in nature?
During this process the residual forces on the surface of adsorbent decreases which in turn decreases the surface energy. This energy appears in the form of heat, hence adsorption is exothermic in nature.Which will be more readily adsorbed on the surface of charcoal ammonia or carbon dioxide Why?
Among CO2 and NH3, NH3 will be more readily absorbed on the surface of the charcoal. This is because the critical temperature of ammonia gas is quite high than the carbon dioxide. Hence, it easily combines with the materials than the carbon dioxide whether it is solid, liquid or any gases.Which has more critical temperature nh3 or co2 and why?
Ammonia molecules are hydrogen bonded to each other while carbondioxide molecules are held together by weak dipole-dipole interactions. NH3 has higher critical temperature and larger VanderWaal's forces of attraction than CO2 and therefore it will be adsorbed more readily than CO2.Why chemisorption first increases with increase in temperature?
Chemisorption initially increases then decreases with rise in temperature. The initial increase is due to the fact that heat supplied acts as activation energy. The decrease afterward is due to the exothermic nature of adsorption equilibrium.Which has higher enthalpy of adsorption physisorption and chemisorption?
Which has a higher enthalpy of adsorption, physisorption or chemisorption? Answer: Chemisorption has a higher enthalpy of adsorption. Because it involves chemical bond formation.What do you understand by activation of adsorbent how is it achieved?
Activation of an adsorbent means, increasing the adsorbing power of the adsorbent. This can be achieved by, making their surface rough by mechanical rubbing or by chemical action. By doing so, it will increase the specific area of the adsorbent.What is the sign of Delta H and Delta S when a gas is adsorbed by an adsorbent?
When a gas is adsorbed , there is a decrease in entropy . Hence, ∆S is positive. There is a decrease in surface energy, hence it appears as heat.So, ∆H is negative.What are the factors which influence the adsorption of a gas on a solid?
- Nature and surface area of adsorbent: Greater the surface area of the adsorbent, greater is the volume of gas adsorbed.
- Temperature: It is observed that the adsorption decreases with the increase of temperature.
- Pressure:
- Activation of the solid adsorbent:
What are the side effects of activated charcoal?
Side effects of activated charcoal include constipation and black stools. More serious, but rare, side effects are a slowing or blockage of the intestinal tract, regurgitation into the lungs, and dehydration.How do you activate charcoal at home?
Making activated charcoal involves heating carbon-rich materials, such as wood, peat, coconut shells, or sawdust, to very high temperatures. This 'activation' process strips the charcoal of previously absorbed molecules and frees up bonding sites again.What is the difference between charcoal and activated charcoal?
Activated charcoal is produced at higher temperatures than charcoal. Activate charcoal is much more porous than charcoal. Activated charcoal is much more effective in filtering material and a more effective adsorbent than charcoal. Activated charcoal is more commonly used in medicine than charcoal.Does activated charcoal adsorb viruses?
Activated carbon is a useful sorbent for removing organics and other spe- cific compounds from water. More re- cently, activated carbon has successfully adsorbed polio virus and infectious hepatitis virus."What happens during activation of charcoal?
Activated carbon, also called activated charcoal, is a form of carbon processed to have small, low-volume pores that increase the surface area available for adsorption or chemical reactions. Further chemical treatment often enhances adsorption properties. Activated carbon is usually derived from charcoal.What drugs Cannot be absorbed by charcoal?
Activated charcoal does not adsorb alcohols (ethanol, methanol), ethylene glycol, iron salts, cyanide, or lithium [25]. However, ethanol does not prevent the adsorption of associated substances.How long does activated charcoal stay in your system?
So, the sooner activated charcoal is taken after swallowing the drug or poison, the better it works—generally within 30 to 60 minutes. The toxic molecules will bind to the activated charcoal as it works its way through your digestive tract, and then they will leave your body together in your stool.Does a sponge adsorb or absorb?
Def. 2a: To suck up or take up such as: a sponge absorbs water, charcoal absorbs gas, and plant roots absorb water. “Adsorption.” Def. 1: The adhesion in an extremely thin layer of molecules (as of gases, solutes, or liquids to the surfaces of solid bodies or liquids with which they are in contact.How do you reactivate charcoal?
Re-Activate the Activated CharcoalStep Two: Take a hair dryer and turn it to high heat. Then dry the inside and outside of the bag thoroughly. The heat passing through the charcoal fabric will reactivate the charcoal lining.